
The most stable isotopes of oxygen are:Įlectronic shells are the energy levels that accommodate electrons. Oxygen has 3 stable isotopes and 18 radioactive isotopes with the shortest half-life. of subatomic particles in the same chemical element. Isotopes are formed when there is variation in the no. The covalent radius of oxygen is 66 pm with an error of +2 or -2 which results in stable diatomic oxygen molecules by double covalent bonding. The van der Waals radius of oxygen is 152 pm as can be judged from its bonding abilities. The boiling point of oxygen is -183 degree Celsius which like its melting point is very low due to the molecular forces between them. The melting point of oxygen is -218 degree Celsius which is quite low because it is covalently bonded which is a weak bonding and does not require much energy. The atomic density of oxygen is 1.429 g/cm 3 which explains the volumetric and spatial capacity of the element. The electronegativity of oxygen according to the Pauling scale is 3.44 which is the second highest which implies high electron attracting capacity as it needs only 2 electrons to complete its octet. Oxygen electronegativity according to Pauling The atomic number of oxygen is 8 because it has 8 protons. Oxygen is situated in the p-block of the periodic table which highlights the non-metallic character and high electron gain enthalpy of oxygen. Oxygen is located in the 2 nd period of the periodic table which follows the decreasing metallic trend and is a non-metal. Oxygen belongs to group 16 of the periodic table which is famously called the chalcogen family. Symbol of diatomic oxygen Oxygen group in the periodic table The symbol for oxygen is O which is the first letter of the element as decided by the atomic theory but in nature it is presented as O 2 which is its diatomic form. Let us analyze oxygen’s periodic classifications and chemical properties. The evolutionary theories regarding the origin of life are very much based on oxygen. Oxygen is the basis of various interdisciplinary sciences and is foundational in the development of organic chemistry and its related studies. 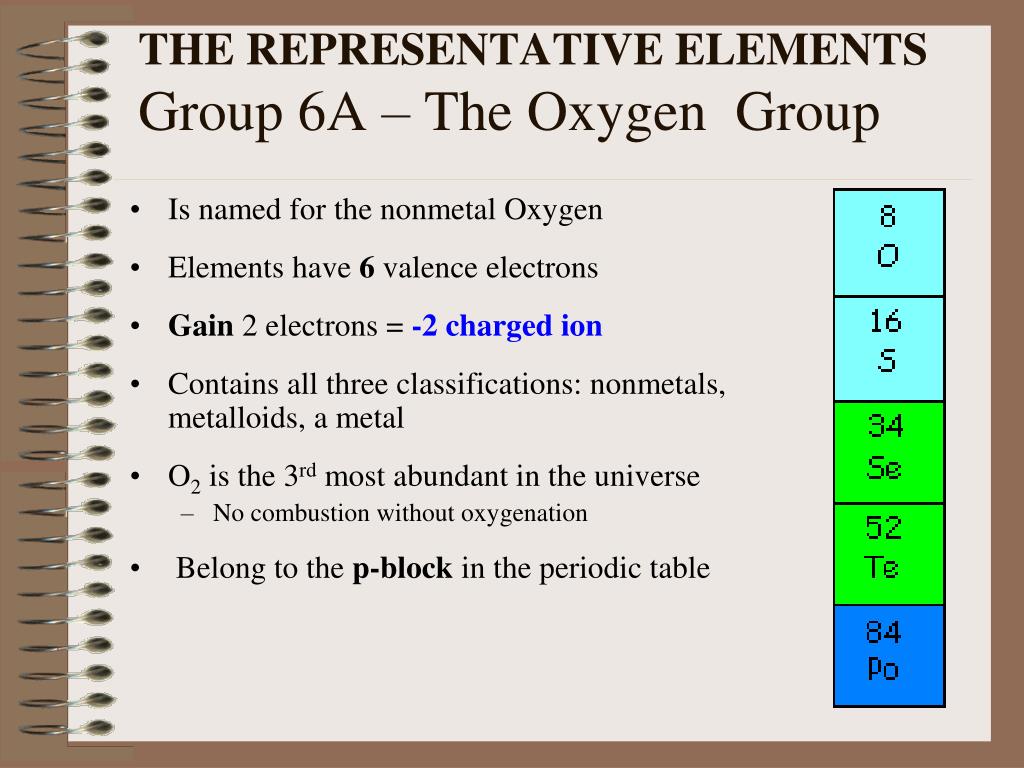
Its existence on earth is in the form of various oxides due to its chemical flexibility and easy binding properties. Oxygen is a diatomic and odorless gas and is a major source of thriving life and other biological and ecological phenomena on the planet earth. Being an element of importance, let us discuss various facts related to it in this article. Oxygen is one of the most significant and abundant elements present on the earth.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
